
“For the first few months, my vision improved with monthly visits and injections. But it’s difficult to have family members take me to my appointments, and I can’t make it to the office that often anymore. My vision is slowly getting worse.”
WHAT IS WET AMD?
Age-related macular degeneration (AMD) is a common degenerative disease of the retina, the light-sensitive tissue in the back of the eye. Wet AMD occurs when the abnormal blood vessels (choroidal neovascularization) grow underneath the macula leading to leakage of fluid and blood leading to visual distortion and acute vision loss. If untreated or undertreated, wet AMD leads to scar formation and permanent damage that results in blindness. Wet AMD is the leading cause of vision loss among individuals 50 years of age and older in the United States and other developed countries.
Are there medicines available to treat wet AMD?
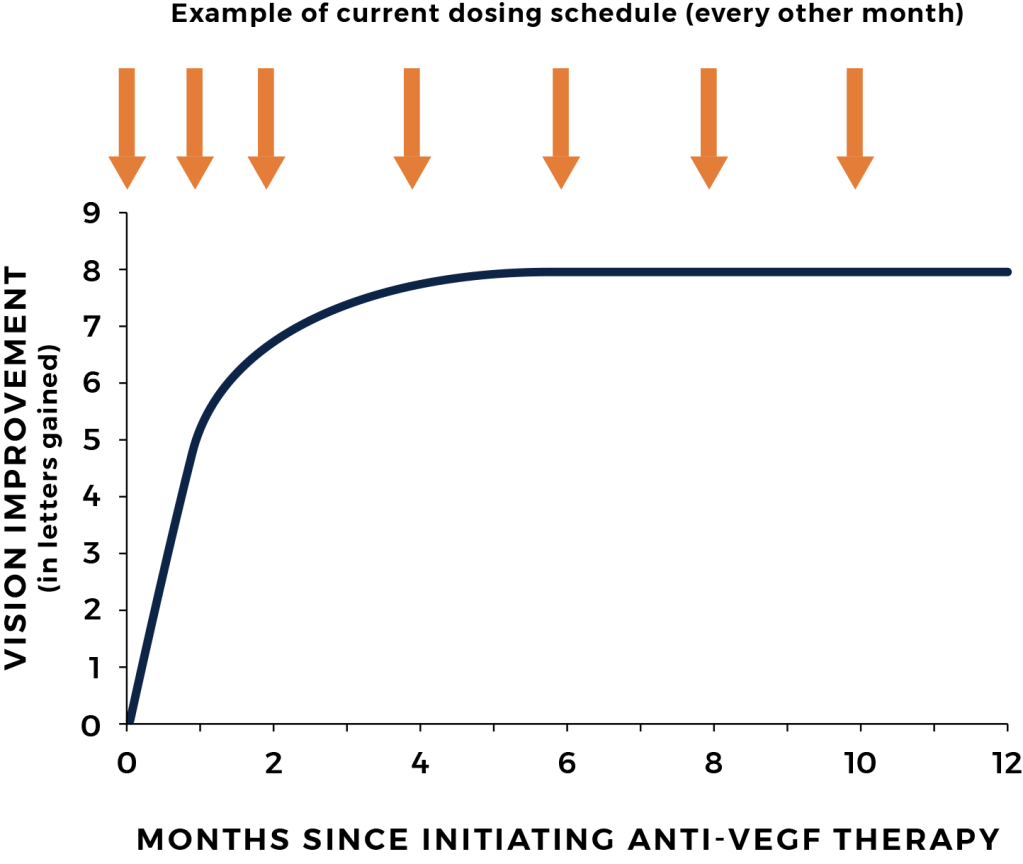
Excess VEGF levels inside the eye play a role in the development of abnormal blood vessels in wet AMD. Current standard of care involves frequent injections in the eye (intravitreal injections), often every 4 to 8 weeks, that block VEGF.
Are these medicines effective?
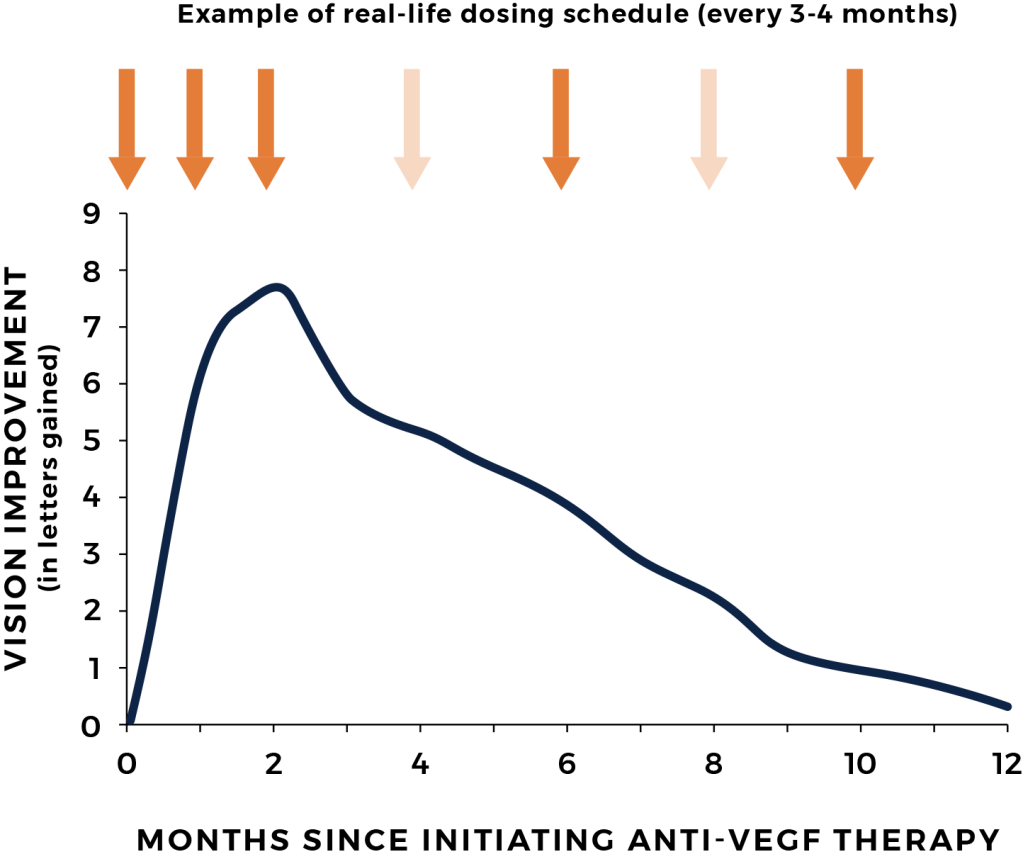
Although intravitreal injections of anti-VEGF agents can be effective, the frequent visits to the doctor’s office are burdensome and difficult to maintain. Non-compliance to these regimens results in vision loss from undertreatment.
Each time the treatment interval is overextended, the patient’s disease can reactivate, which leads to incremental and cumulative damage to the retina. Over time this may lead to permanent loss of vision.
HOW TARCOCIMAB TEDROMER (KSI-301) CAN HELP
Our therapeutic candidate tarcocimab tedromer is a novel anti-VEGF biologic built on our propriety antibody biopolymer conjugate (ABC) platform.
Tarcocimab tedromer is administered as an intravitreal injection and designed to provide sustained inhibition of VEGF for up to 6 months. Primary results of the Phase 2b/3 DAZZLE study, evaluating tarcocimab tedromer’s efficacy, durability and safety, are now available here.
