GENERATION 2.0
Inspired to build the world’s leading high-science retina pipeline
OUR PIPELINE
Our pipeline is designed to address key limitations of today’s therapies across a broad spectrum of retinal diseases.
BLA-ready profile in wet AMD, DR and RVO
DAYBREAK (wet AMD) enrollment complete
DAYBREAK (wet AMD) enrollment complete
PEAK and PINNACLE actively recruiting

OUR CANDIDATES
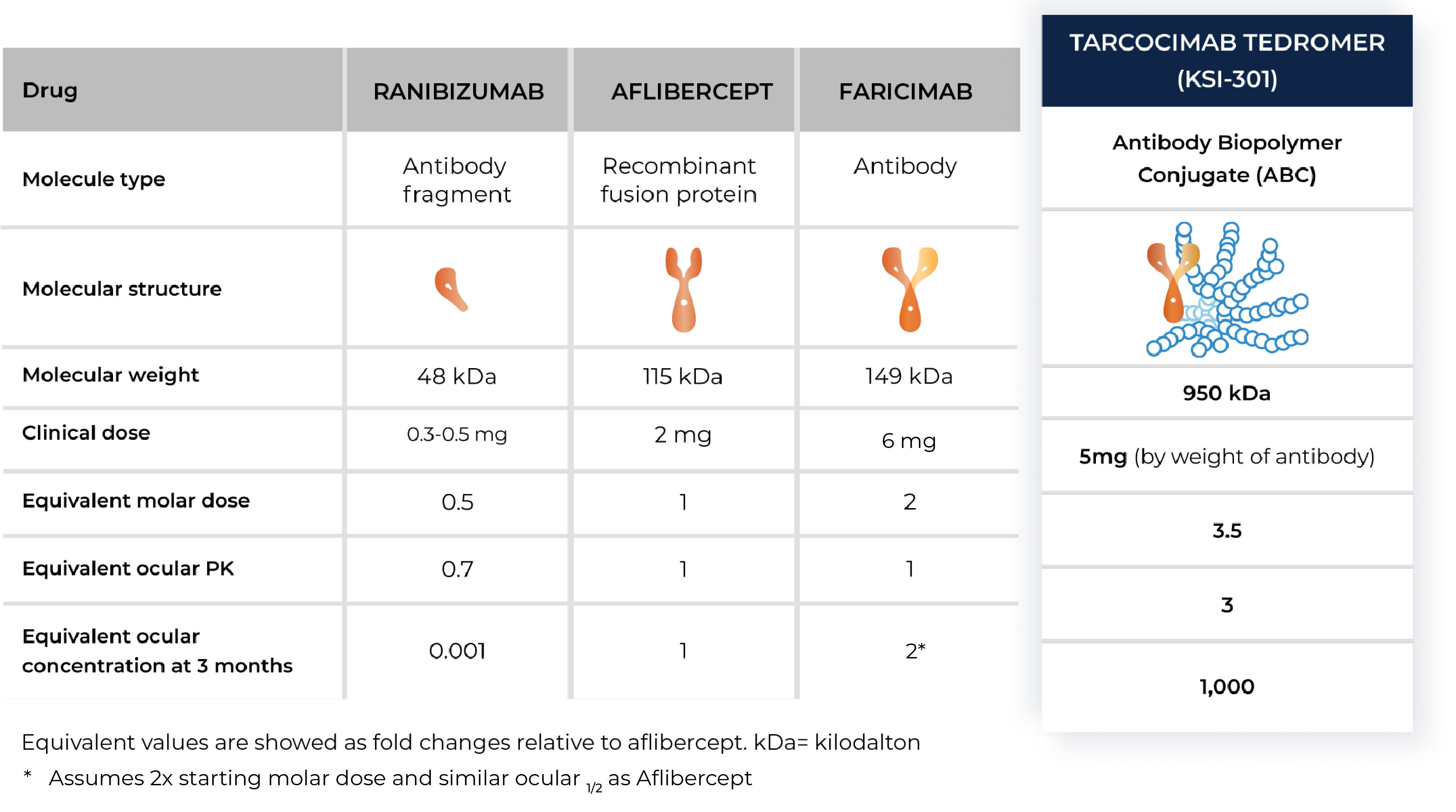
Zenkuda™ (tarcocimab)
Anti-VEGF
KSI-501
Anti-IL-6, VEGF trap

KSI-101
Anti-IL-6, VEGF trap

Multi-mechanistic medicines
1 Molecule, Many targets
An investigational anti-VEGF biologic designed
to deliver immediate and durable disease control
Zenkuda (tarcocimab tedromer) is our most advanced program, supported by four successful Phase 3 studies.
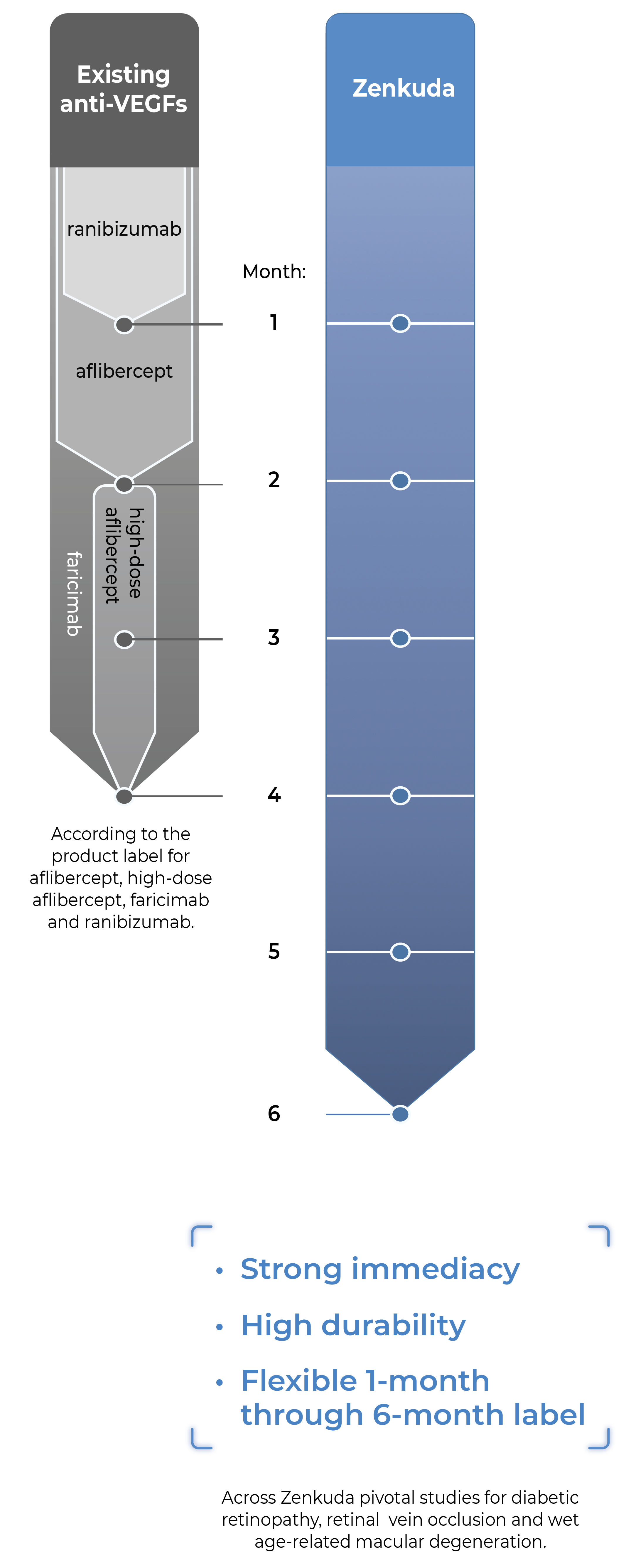
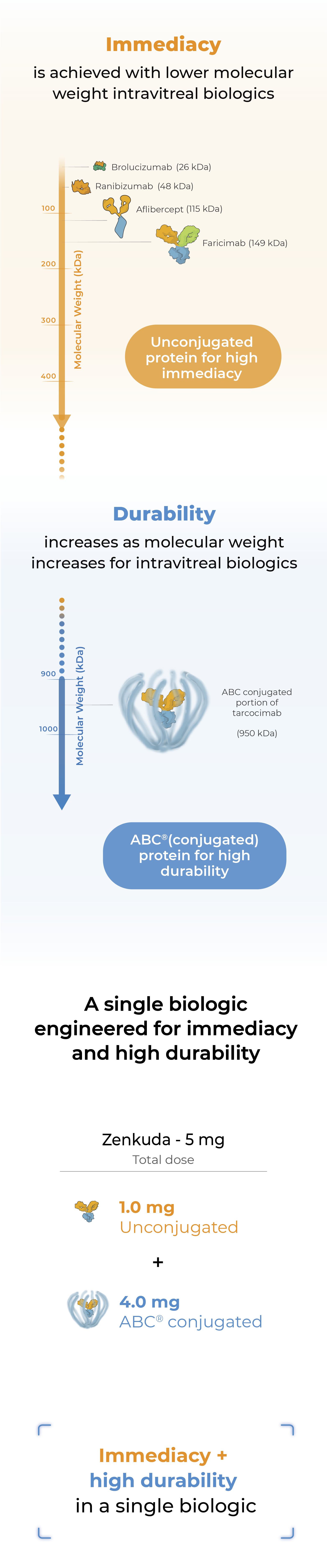
Zenkuda is an anti-VEGF Antibody Biopolymer Conjugate (ABC®) being developed to provide strong immediacy and high durability. Across multiple studies in high-prevalence retinal vascular diseases, Zenkuda demonstrated consistent 6-month predominant durability and favorable safety. Designed for flexible dosing from 1 to 6 months, Zenkuda has the potential to be a mainstay intravitreal biologic for a broad range of patients.

- Across Zenkuda pivotal studies for diabetic retinopathy, retinal vein occlusion and wet age-related macular degeneration.
- According to the product label for aflibercept, high-dose aflibercept, faricimab and ranibizumab.
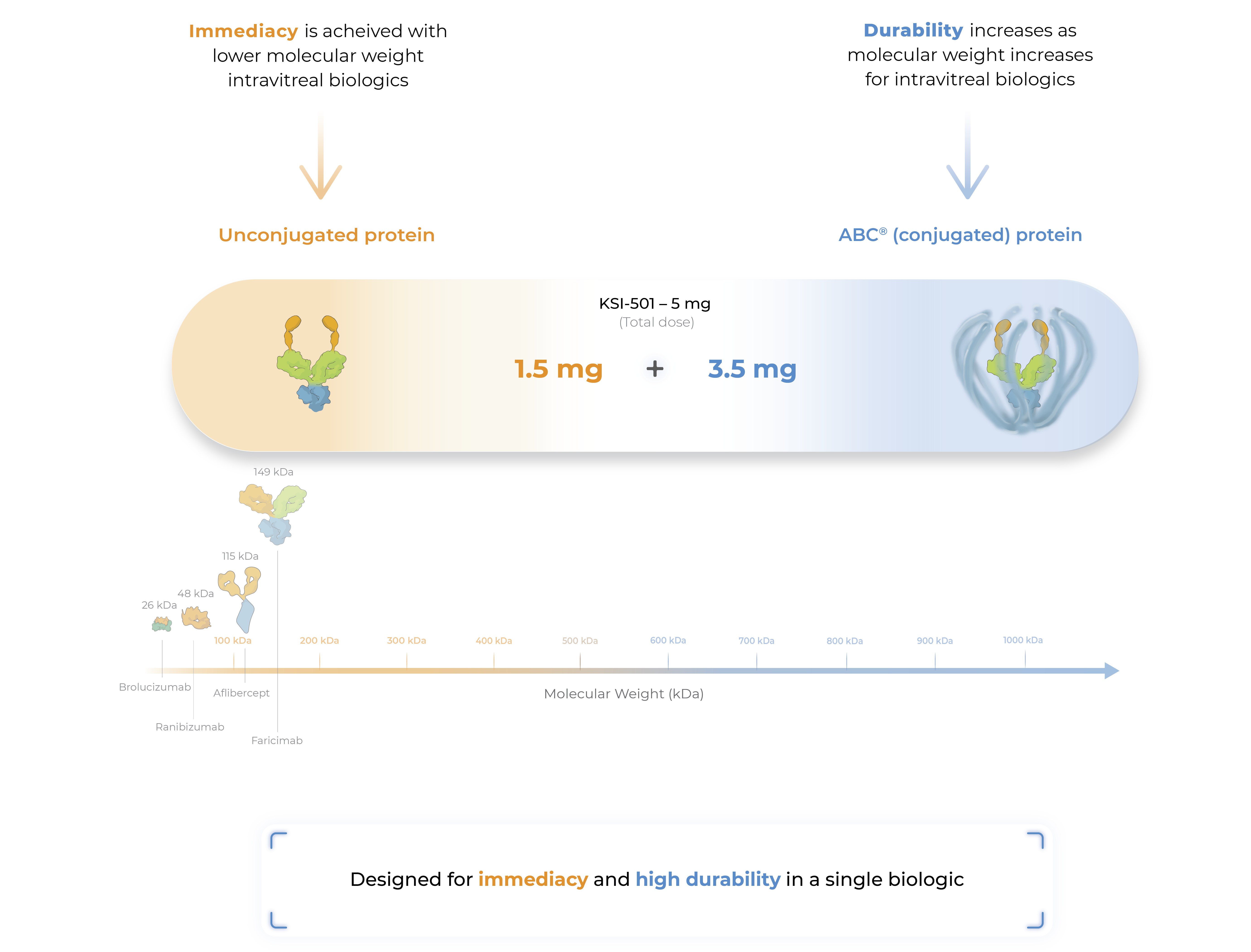
Zenkuda combines immediacy and high durability in a single biologic
Zenkuda combines unconjugated and ABC®(conjugated) protein in a single biologic. The unconjugated protein is designed to deliver a strong “pulse” of VEGF inhibition (immediacy) and the conjugated protein is designed to persist in the eye to provide sustained disease control.

Tarcocimab and the Science of Durability
Tarcocimab is an ABC medicine built to last. This designed-in durability is supported by clinical data and is what we call our science of durability. See the data
Zenkuda and the Science of Durability
Zenkuda is an ABC medicine built to last. This designed-in durability is supported by clinical data and is what we call our science of durability. See the data
Four successful Phase 3 studies across major retinal diseases
Primary endpoint met and compelling durability demonstrated.
| Study Design | Primary Endpoint | Extended Durability | ||
|---|---|---|---|---|
| GLOW1 Study Diabetic Retinopathy |
|
✓ | ✓ | See Results |
| GLOW2 Study Diabetic Retinopathy |
|
✓ | ✓ | See Results |
| BEACON Study Retinal Vein Occlusion |
|
✓ | ✓ | See Results |
| DAYLIGHT Study Wet Age-Related Macular Degeneration |
|
✓ | Not Applicable | See Results |
* After treatment initiation phase.
One Phase 3 clinical study ongoing.
| Study Design | Primary Endpoint | Anticipated Timing | ||
|---|---|---|---|---|
| DAYBREAK Study Wet Age-Related Macular Degeneration |
|
Topline data expected 3Q 2026 | See Results |
Our objective is for Zenkuda to be a mainstay intravitreal biologic monotherapy that provides high immediacy and high durability for all patients
Phase 3 results in diabetic retinopathy
Zenkuda demonstrated strong efficacy and differentiated durability across the GLOW1 and GLOW2 studies in diabetic retinopathy
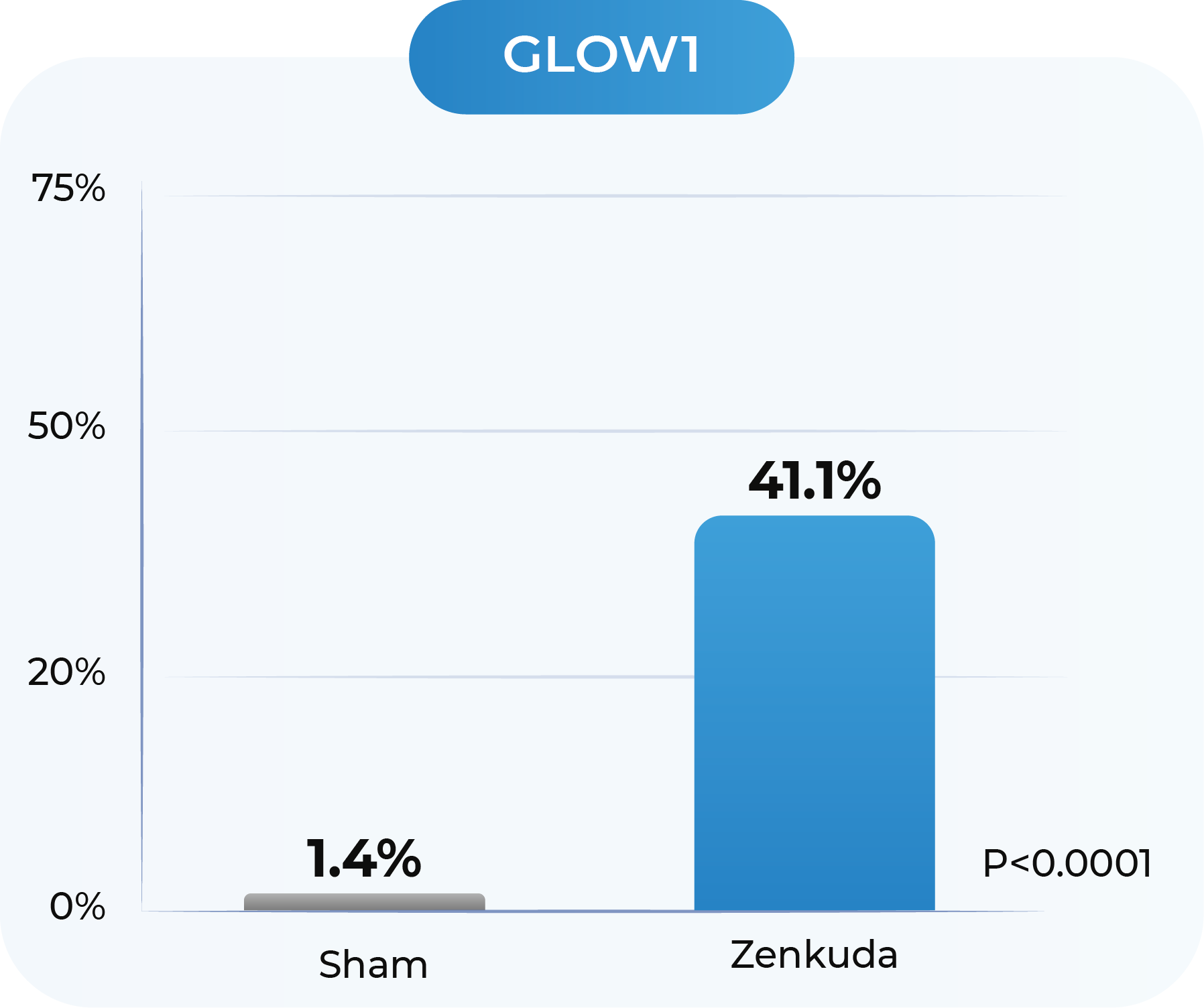
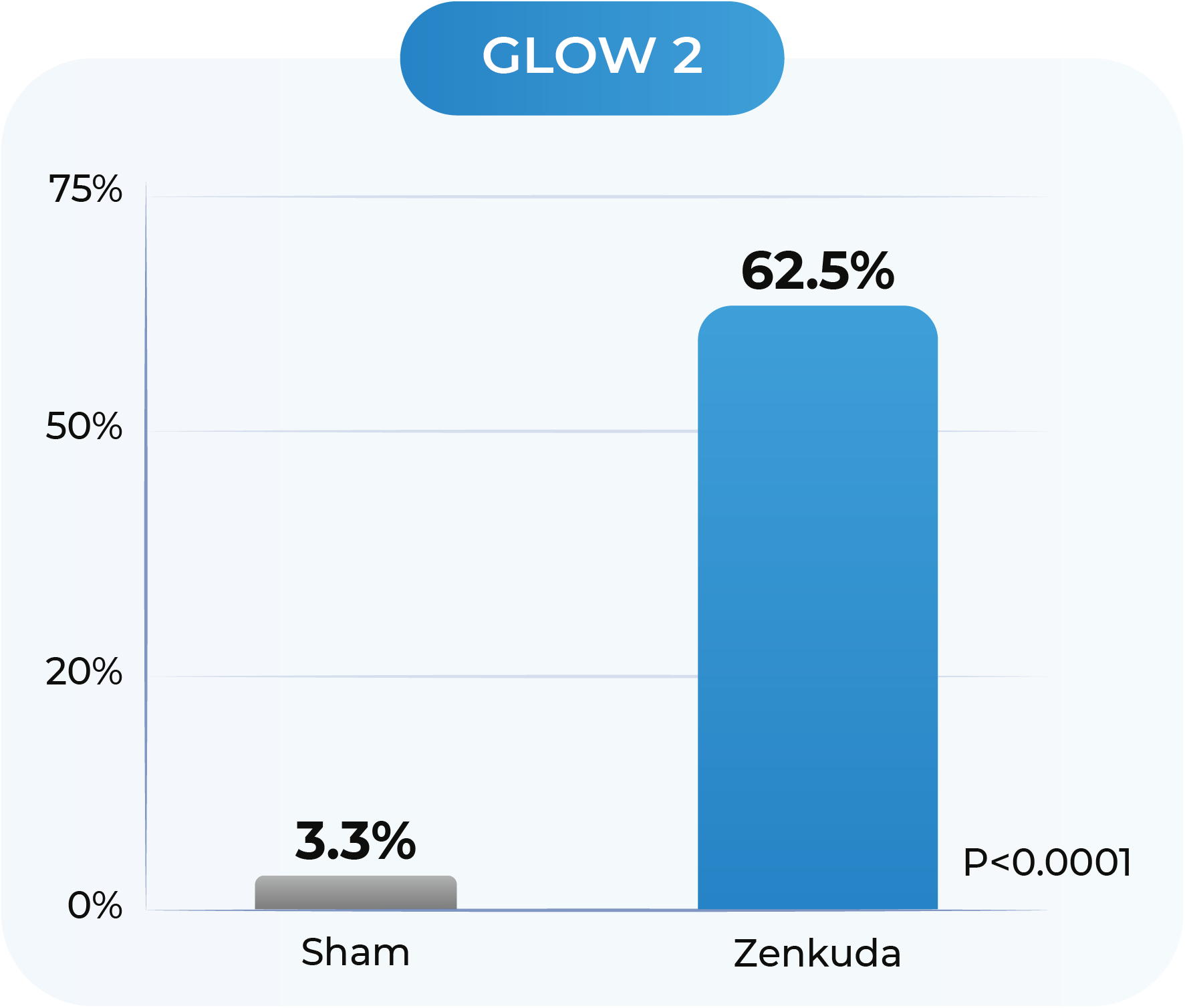
GLOW1 and GLOW2: Proportion of patients with ≥2-Step improvement in DRSS from Baseline to Week 481

GLOW1: Sham (n=125); Zenkuda (n=128). GLOW2: Sham (n=125); Zenkuda (n=130). Week 48 (LOCF) represents the last available observation while on randomized treatment, within the Week 48 visit window. Note: Weighted percentages are based on weighted average of observed estimates across strata using CMH weights. p-values are based on the difference in response rates.
All patients treated with Zenkuda were on a 6-month dosing interval
(after treatment initiation phase)
Zenkuda demonstrated superiority in ≥2-step and ≥3-step improvement in DRSS
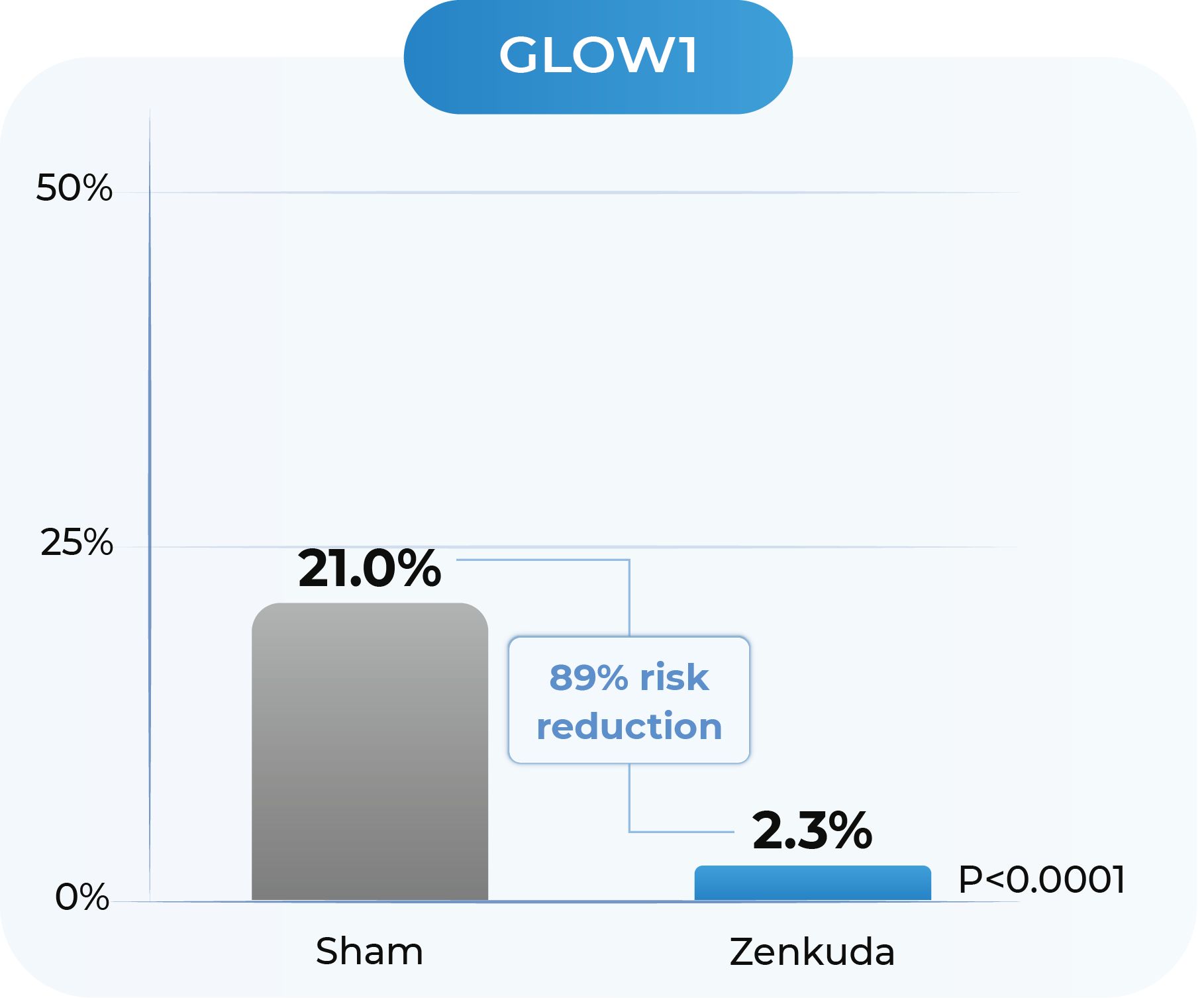
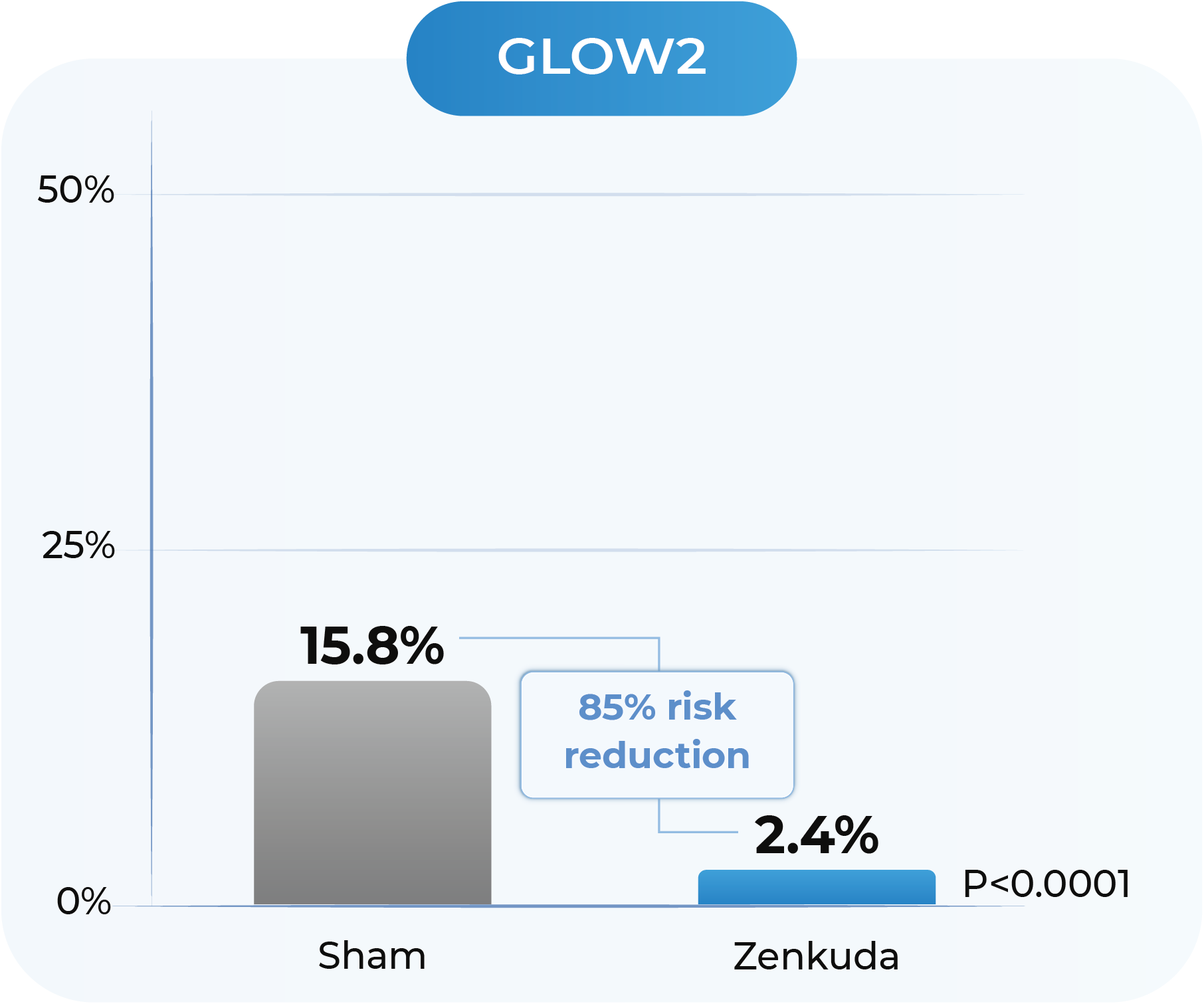
GLOW1 and GLOW2: Proportion of patients developing any sight-threatening complication from Baseline to Week 48

GLOW1: Sham (n=125), Zenkuda; (n=128). GLOW2: Sham (n=125), Zenkuda (n=130). Weighted percentages are based on weighted average of observed estimates across strata using CMH weights. p-values are based on the difference in response rates. Sight threatening complications include: diabetic macular edema, new or worsening proliferative diabetic retinopathy; anterior segment neovascularization; neovascularization of the disc and elsewhere, vitreous hemorrhage and neovascular glaucoma.
In GLOW1 and GLOW2, Zenkuda reduced the risk of developing a pre-specified sight-threatening complication by ≥85%
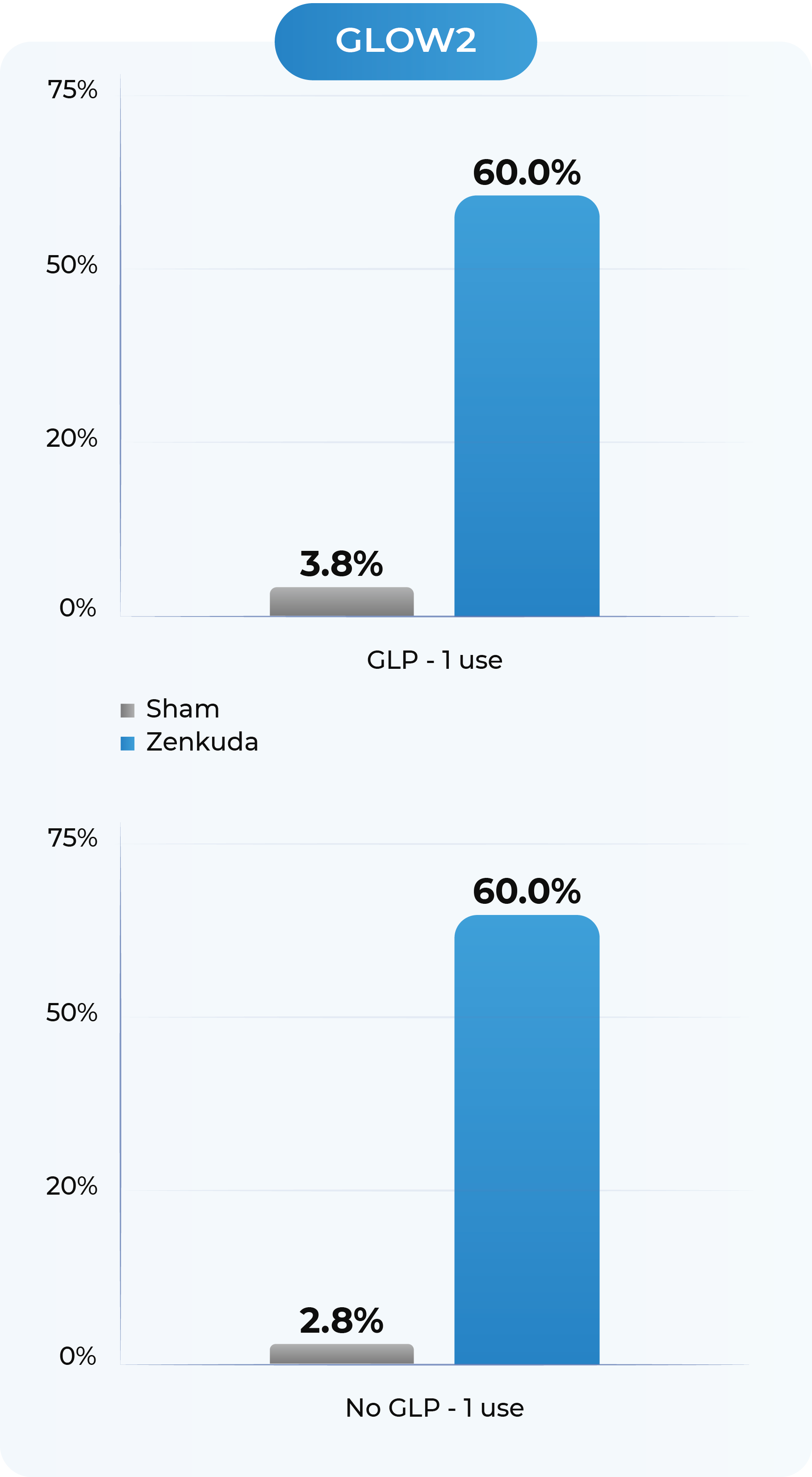
GLOW2: Proportion of patients with a ≥2-Step improvement in DRSS from Baseline to Week 48, by concomitant GLP-1 medication use

Sham GLP-1 use (n=53); Sham No GLP-1 use (n=72). Zenkuda GLP-1 use (n=60); Zenkuda no GLP-1 use (n=70); Note: Percentages are 100*n/N. Week 48 (LOCF) represents the last available observation while on randomized treatment, within the Week 48 visit window.
Zenkuda improved the DRSS score irrespective of the use of GLP-1 medications, showing promising efficacy for real-world use
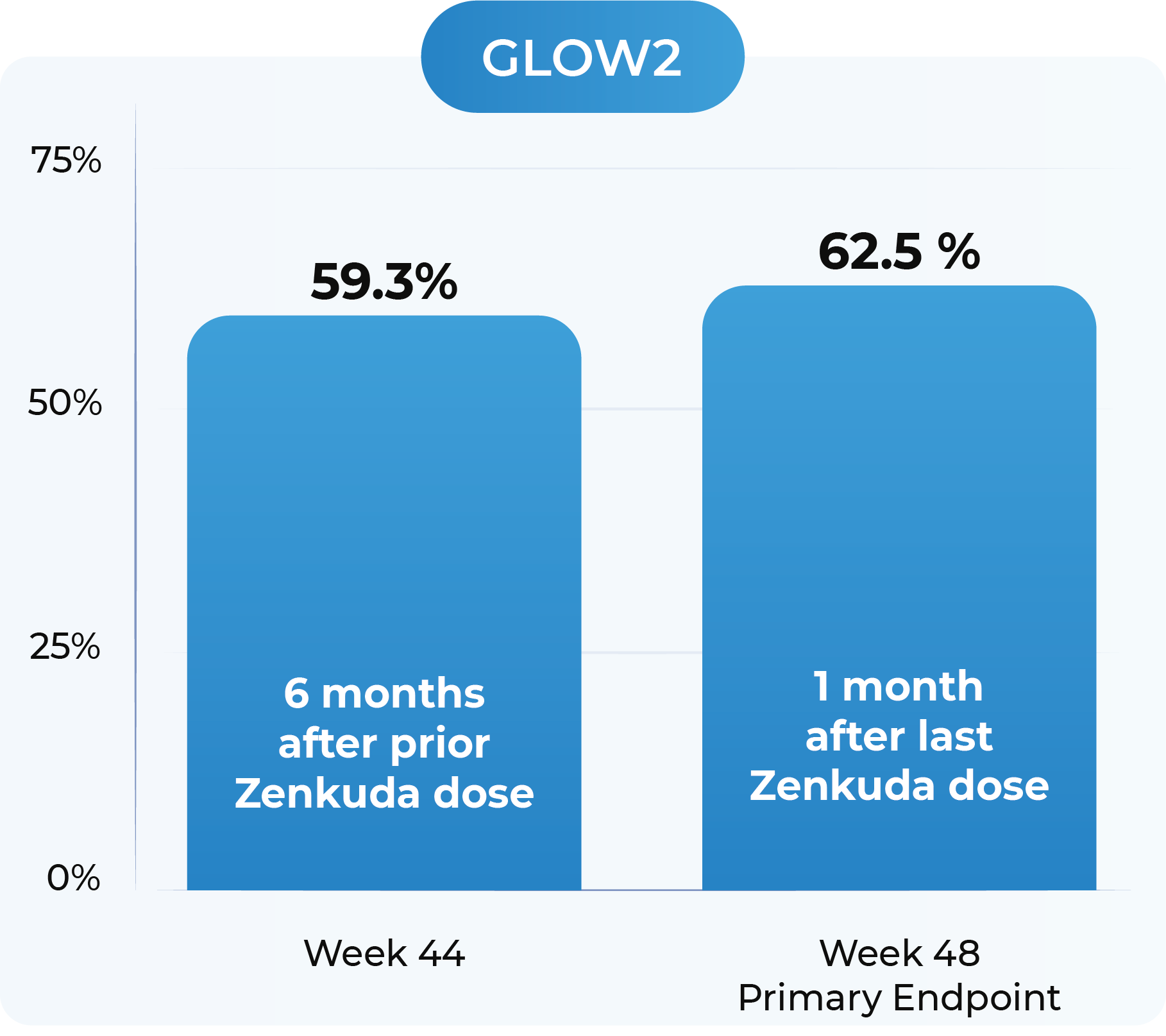
GLOW2: Proportion of patients with a ≥2-Step improvement in DRSS from Baseline to Week 44 and Week 48

GLOW2: Zenkuda (n=130). Week 44 and Week 48 (LOCF) represents the last available observation while on randomized treatment, within the Week 44 and Week 48 visit windows, respectively. Weighted percentages are based on weighted average of observed estimates across strata using CMH weights.
Zenkuda demonstrated robust ≥2-step improvement in DRSS at the 6-month dosing interval
Phase 3 results in retinal vein occlusion
In the Phase 3 BEACON study, Zenkuda dosed every 8 weeks was non-inferior to aflibercept dosed every 4 weeks during the first 6 months. In the second 6 months, Zenkuda demonstrated 6-month durability in 75% of patients, with consistent visual and anatomical gains.
BEACON: Treatment distribution through Week 48
78% of patients received ≤5 doses with Zenkuda
93% of patients received ≥6 doses with aflibercept

Zenkuda demonstrated a reduction in treatment burden through 48 weeks for both BRVO and all RVO patients vs aflibercept
BEACON: Mean BCVA and CST from Baseline to Week 48

Mean observed data; Week 24 and 48 datapoints are Mean (Standard Deviation). Results for BCVA are based on a mixed model repeated measures (MMRM) analysis, with the change from baseline value as the dependent variable; treatment, visit (Week 1 through Week 48), and treatment by visit interaction as fixed effects; randomization stratification variables [baseline BCVA, disease duration, RVO type) and geographical location], as well as continuous covariates of baseline BCVA value and baseline OCT CMM value, as fixed effects; and subject as a random effect
Fixed dosing: Zenkuda achieved similar visual and anatomical gains with two less doses (4 vs 6, respectively)
Individualized dosing: In 75% of patients, Zenkuda delivered 6-month durability with similar visual and anatomical gains from Week 24 to 48
1. GLOW1 and GLOW2 were prospective, randomized, double-masked, sham-controlled, multicenter Phase 3 studies evaluating Zenkuda 5mg in participants with diabetic retinopathy. Both studies employed extended-interval dosing regimens with an ultimate treatment interval of every six months. The primary endpoint was the proportion of eyes improving by ≥2 steps on the DRSS from baseline at Week 48. Additional outcome measures include the proportion of eyes developing a sight-threatening complication of diabetic retinopathy and the proportion of eyes improving ≥3 steps on DRSS from baseline at Week 48. Additional information about GLOW1 and GLOW2 can be found on www.clinicaltrials.gov under Trial Identifier NCT05066230 (https://clinicaltrials.gov/study/NCT05066230) and NCT06270836 (https://clinicaltrials.gov/show/NCT06270836).
2. The Phase 3 BEACON study is a global, multi-center, randomized study designed to evaluate the durability, efficacy and safety of tarcocimab tedromer Q8W vs. aflibercept Q4W in patients with macular edema due to RVO.
DRSS: diabetic retinopathy severity scale
First-in-class
An investigational anti-IL-6 and VEGF trap bispecific biologic designed for higher efficacy and higher durability
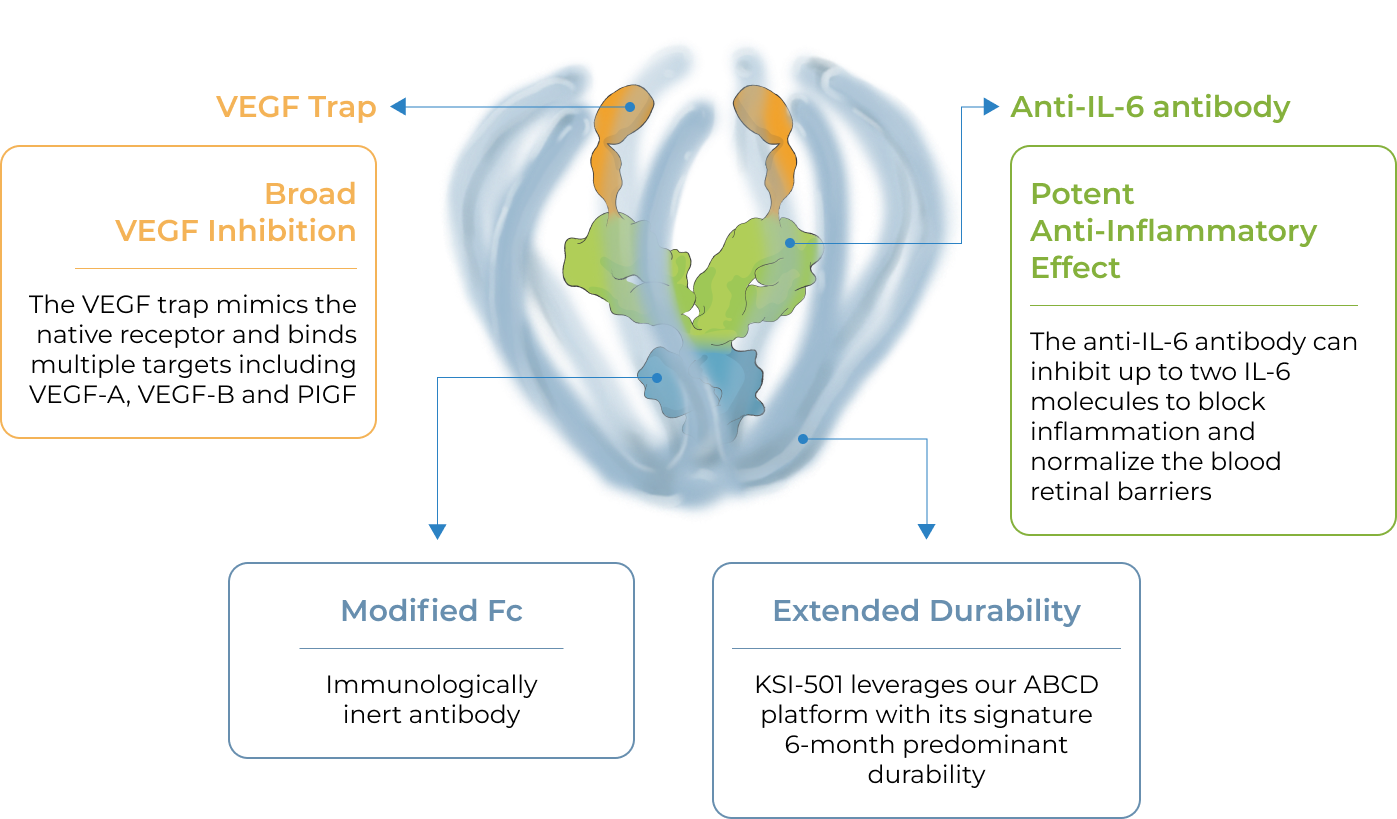
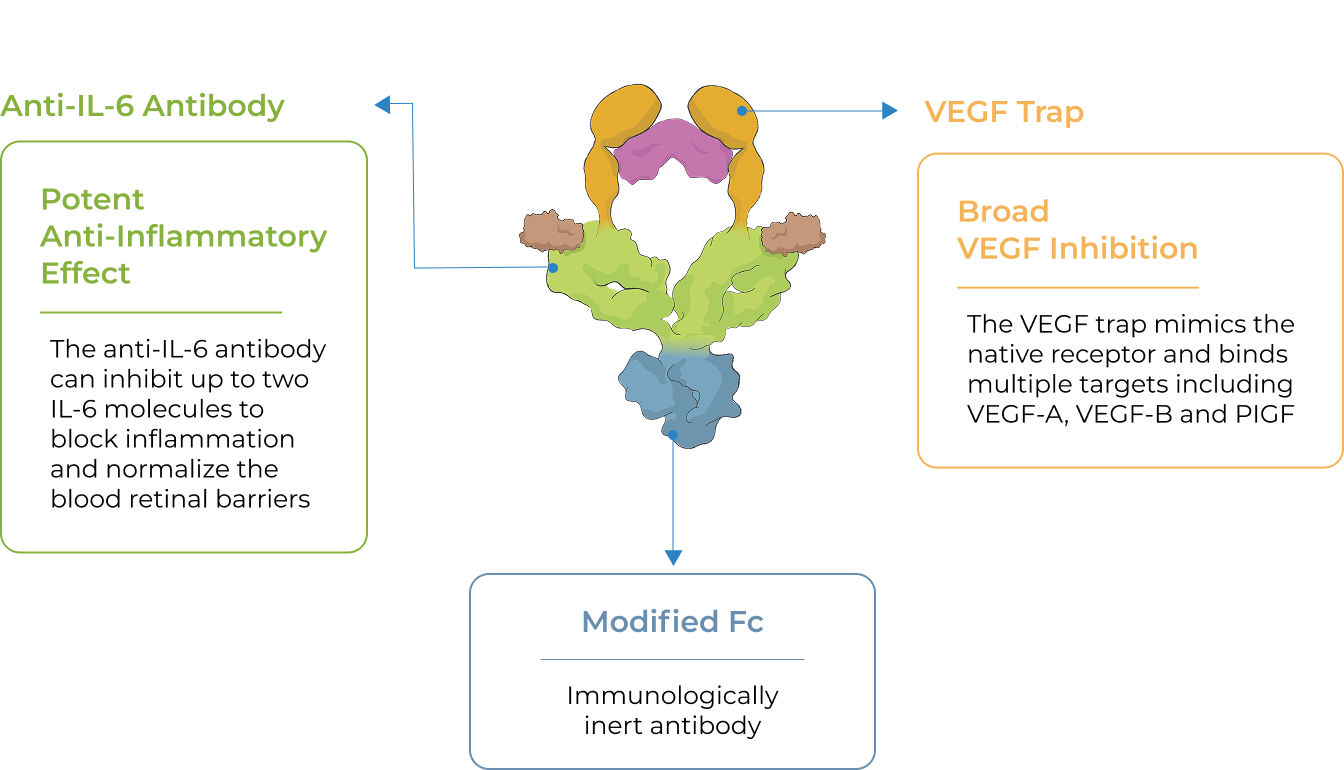
KSI-501 is a bispecific Antibody Biopolymer Conjugate (ABC®) designed to address two key unmet needs in high-prevalence retinal vascular diseases – higher efficacy and higher durability – by targeting retinal inflammation and vascular permeability simultaneously.
Inflammation has been shown to play a significant role in high-prevalence retinal vascular diseases. However, no treatments exist that concurrently address vascular permeability and inflammation.
KSI-501 is designed to inhibit VEGF and IL-6, a pro-inflammatory cytokine and immune growth factor, combining two powerful mechanisms of action to address retinal vascular disease and the underlying inflammatory cascade.
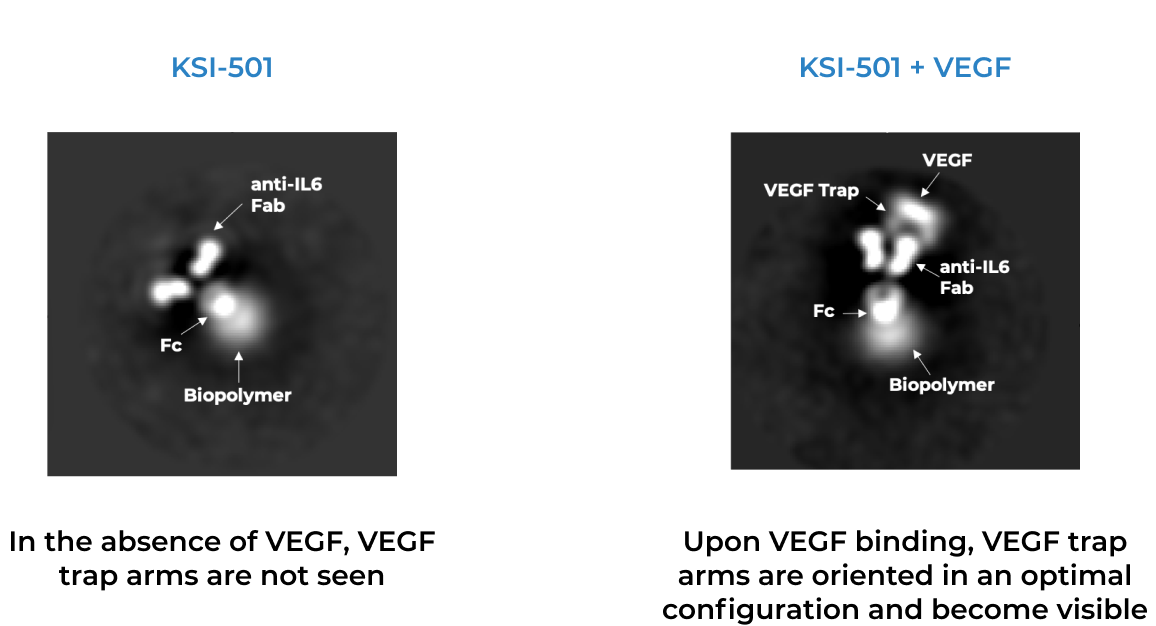
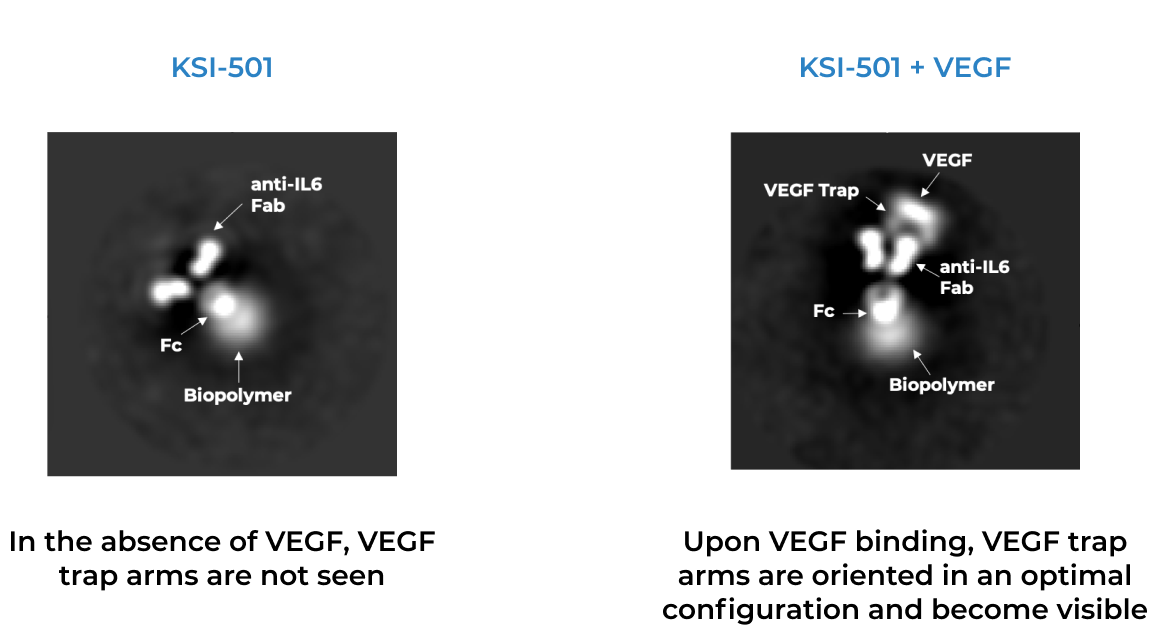
KSI-501 is engineered for highly efficient binding to both IL-6 and VEGF
The anti-permeability effect of VEGF inhibition is the primary effector, with the anti-inflammatory effect of IL-6 inhibition offering the potential for additional clinical benefits.
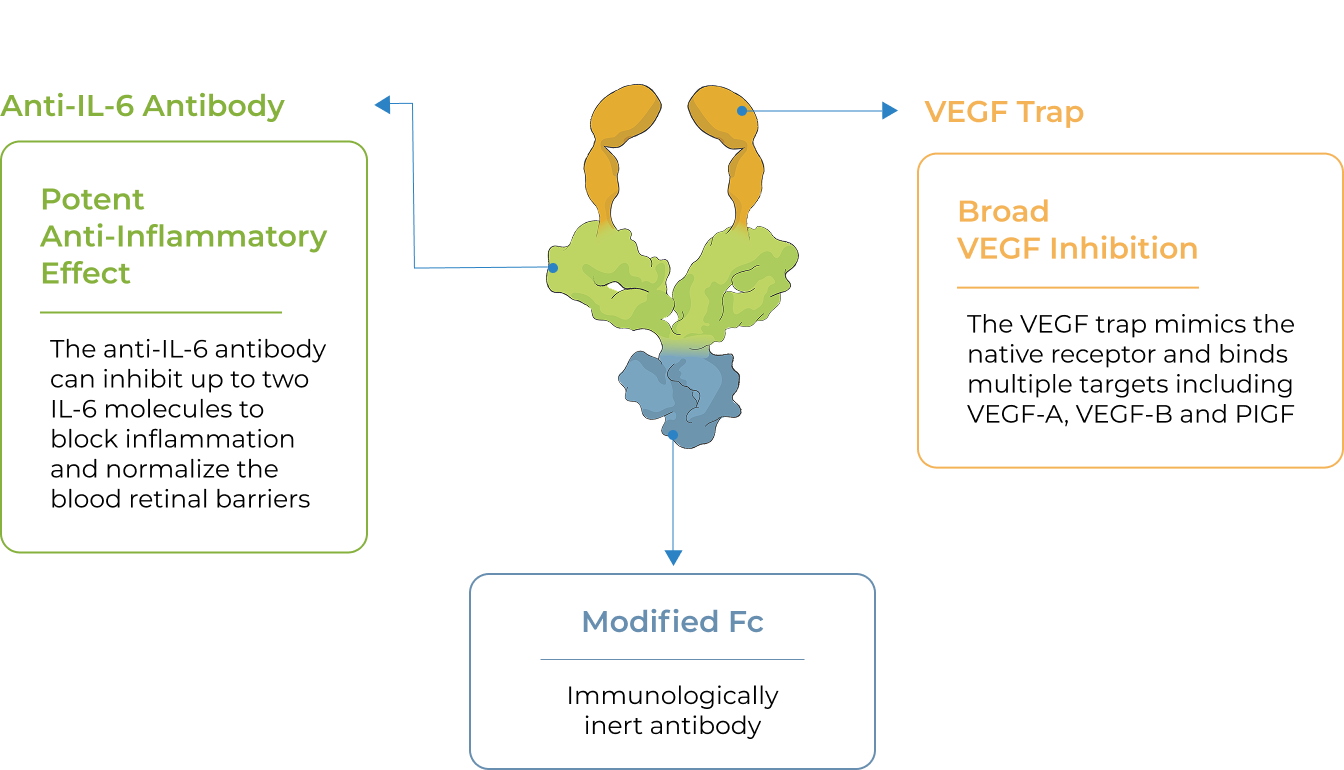
KSI-501 combines immediacy and high durability in a single biologic
KSI-501 combines unconjugated and conjugated protein in a single biologic. The unconjugated protein is designed to deliver a strong “pulse” of IL-6 and VEGF inhibition and the conjugated protein is designed to persist in the eye to provide sustained disease control.
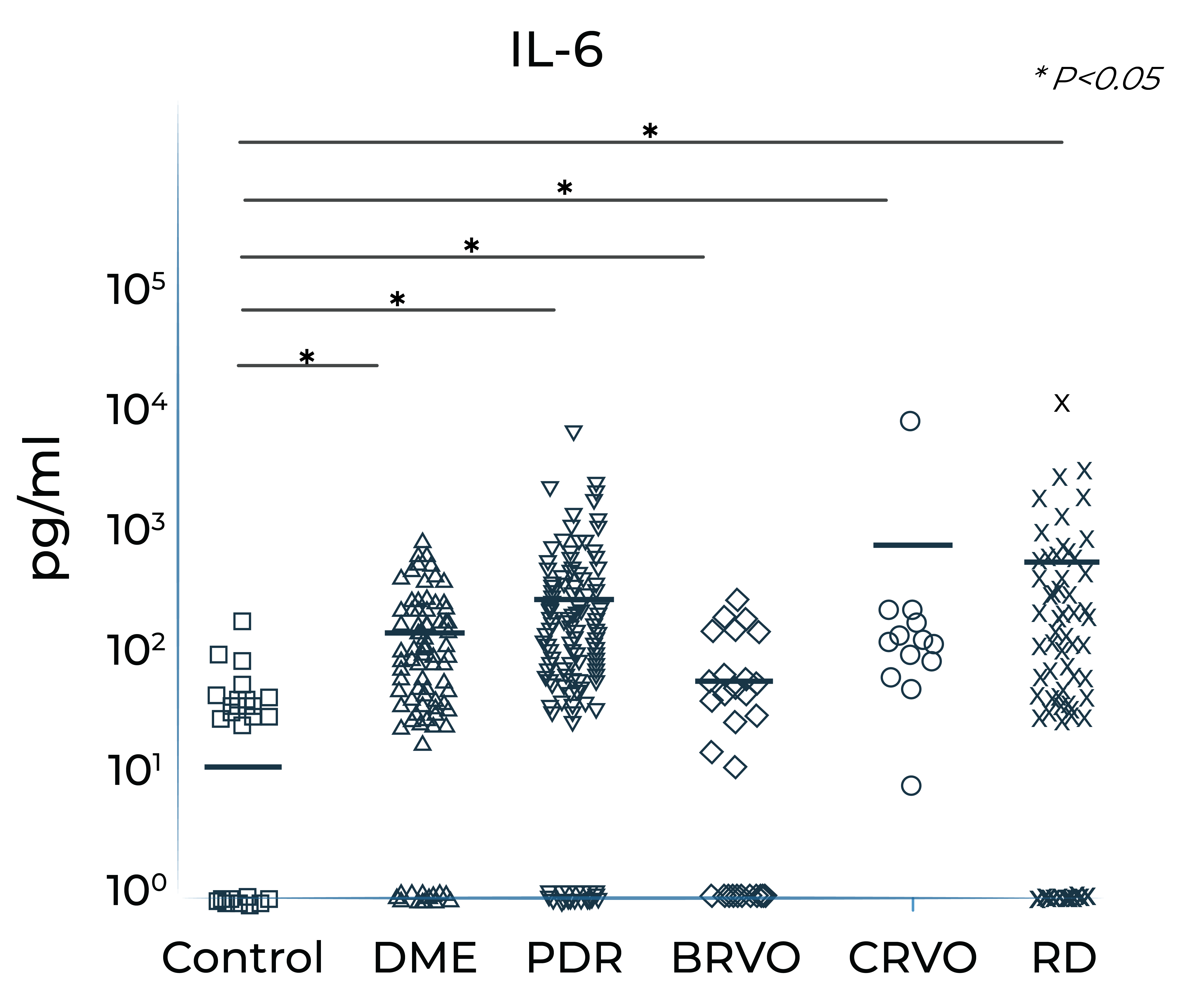
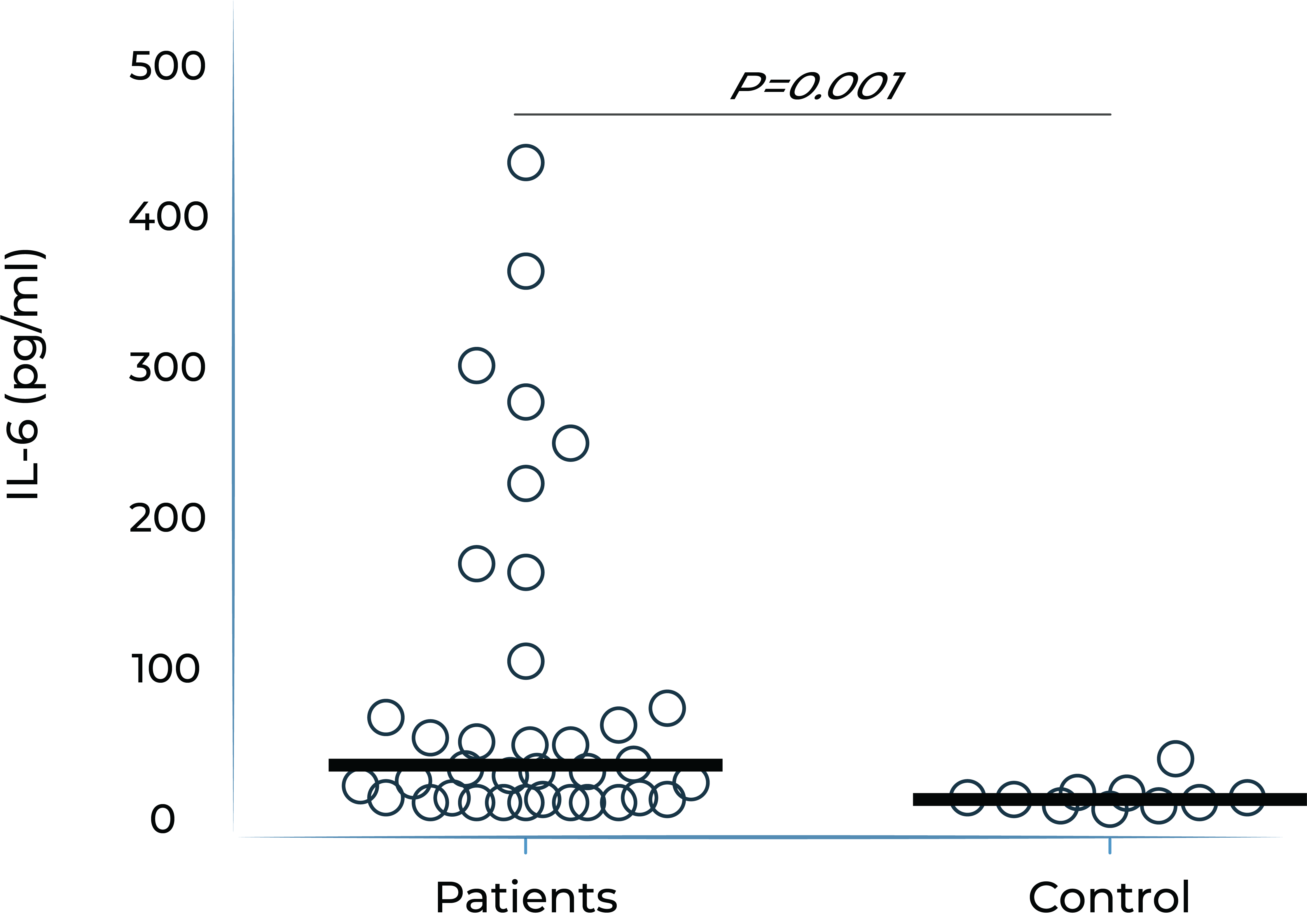
IL-6 levels are significantly elevated in eyes with retinal vascular disease and are implicated in anti-VEGF treatment resistance
Vitreous IL-6 levels in patients with retinal vascular disease vs. control1

DME: diabetic macular edema; PDR: proliferative diabetic retinopathy; BRVO: branch retinal vein occlusion; CRVO: central retinal vein occlusion; RD: retinal detachment.
1.Yoshimura et al. (2009). PLoS ONE 4(12): e8158.
Vitreous IL-6 levels are significantly elevated in patients with retinal vascular diseases
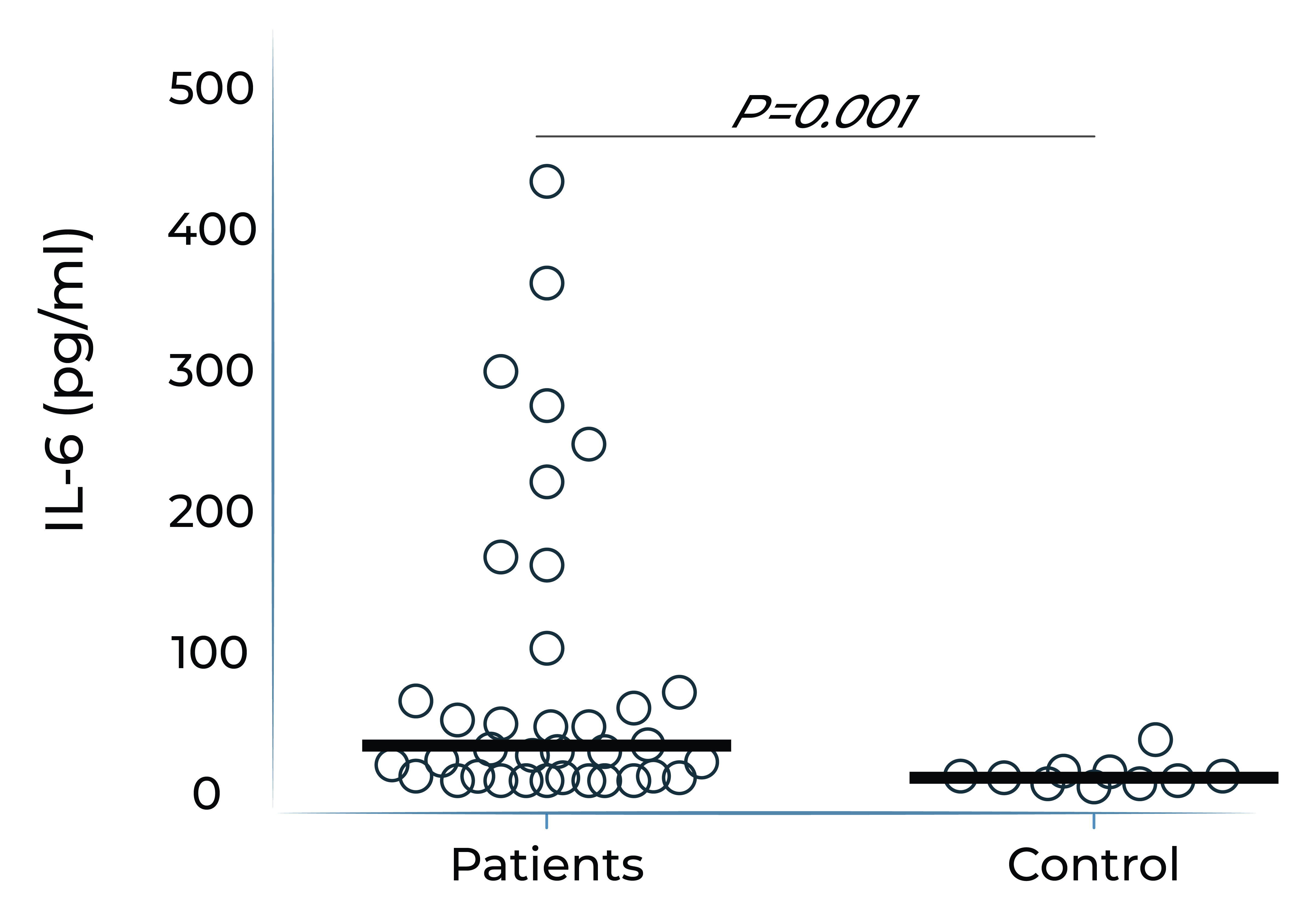
Aqueous humor IL-6 levels in patients with wet AMD at baseline and after anti-VEGF treatment2
Patients that respond to anti-VEGF

Anti-VEGF treatment resistant patients

- Adapted from Chalam et al. (2014). Journal of Ophthalmology, Article ID 502174. Mean with SEM plotted.
Higher aqueous humor IL-6 levels in wet AMD and DME patients treated with anti-VEGF monotherapy correlate with poorer best corrected visual acuity (BCVA) outcomes over time
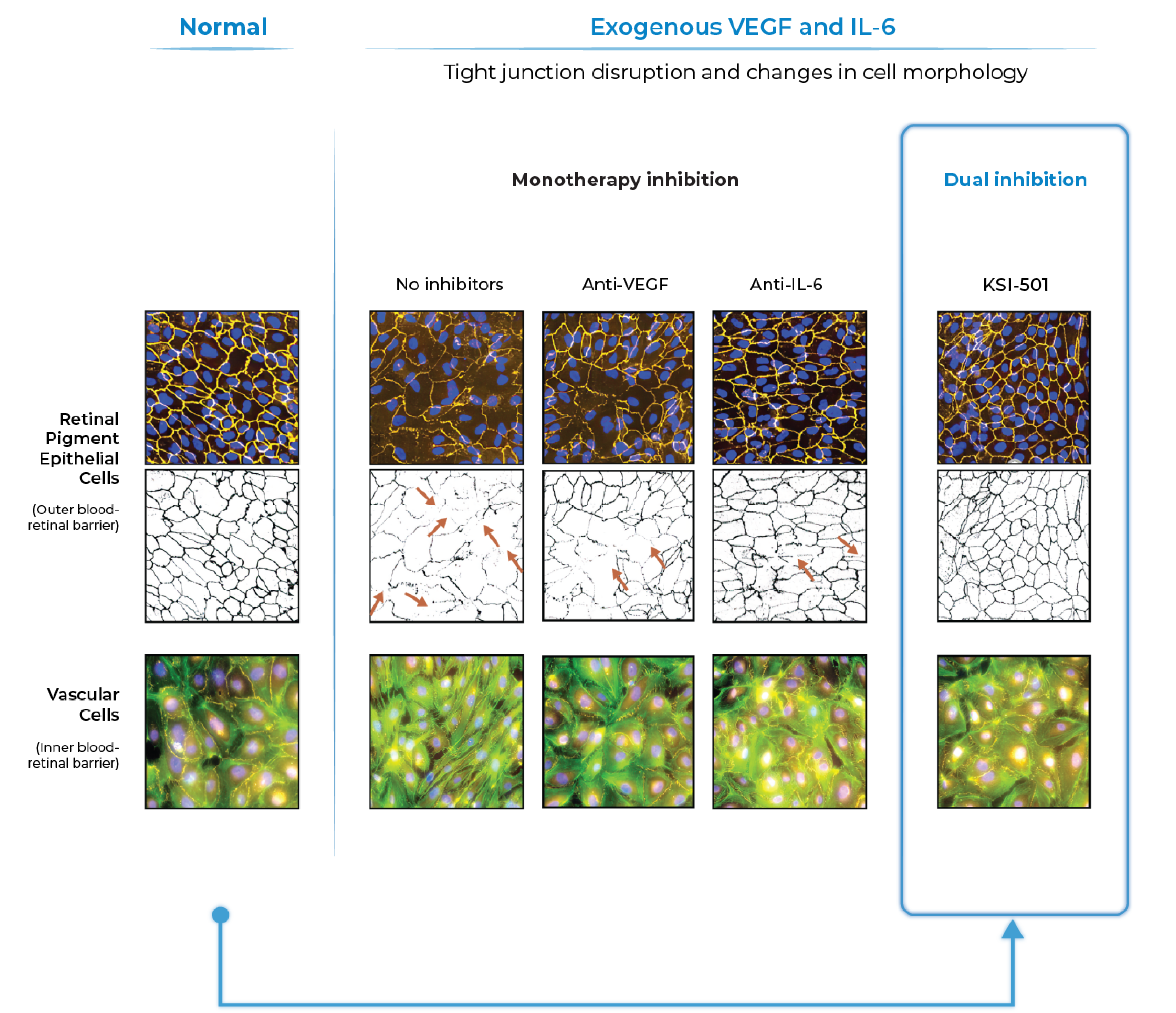
In pre-clinical studies, dual inhibition of IL-6 and VEGF by KSI-501 show a synergistic effect on barrier biology
Dual inhibition of VEGF and IL-6 by KSI-501 confers superior normalization of complex tight junction-mediated barrier biology compared to either anti-VEGF or anti-IL-6 monotherapy alone demonstrating the synergistic effect of IL-6 and VEGF dual inhibition on retinal vascular disease.

Dual inhibition of VEGF and IL-6 by KSI-501 confers superior normalization of complex tight junction-mediated barrier biology compared to either anti-VEGF or anti-IL-6 monotherapy alone
With dual effect on the blood retinal barrier, KSI-501 holds the potential to be a new disease-modifying therapy
Phase 3 DAYBREAK study of KSI-501 in wet AMD
KSI-501 is being investigated in the ongoing Phase 3 DAYBREAK study in wet AMD, designed to explore the efficacy potential of bispecific IL-6 and VEGF inhibition in fixed Q8W with individualized monthly dosing of KSI-501.
DAYBREAK Study
Wet Age-Related
Macular Degeneration
- Designed as a registrational study for both tarcocimab and KSI-501
- Tarcocimab objective: assess 6-month durability potential with individualized Q4W-Q24W dosing
- KSI-501 objective: explore efficacy potential of bispecific IL-6 and VEGF inhibition in fixed Q8W dosing with additional individualized monthly dosing
- Uses enhanced 50 mg/mL formulation
In wet AMD, there is preclinical evidence that IL-6 is implicated in the development of choroidal neovascularization and clinical evidence demonstrating that IL-6 is associated with development and progression of AMD, resistance to anti-VEGF treatment in wet AMD, and reactivation of disease by promoting growth of new neovascular membranes.
DAYBREAK follows the encouraging results of the Phase 1 study of KSI-501 in DME, a disease known to have high levels of cytokine-mediated microvascular inflammation in addition to VEGF-mediated vascular permeability.
Phase 1
Diabetic Macular
Edema
- 16 patients
- Multiple ascending dose design
- Each patient received 3 monthly doses (Day 1, Week 4 and Week 8) and was followed for 24 weeks total
KSI-501 has three tiers of innovation
Two-target
mechanism of action
![]()
Designed to address two key unmet needs: higher efficacy and higher durability by inhibiting the IL-6 inflammation pathway and the dominant VEGF pathway
Enhanced
formulation
![]()
Enhanced 50 mg/mL formulation is designed to deliver both strong immediacy and high durability
Science of
durability
![]()
Supported by our science of durability
First-in-class
An investigational, high-strength, bispecific protein designed to treat macular edema secondary to inflammation (MESI) for which no approved intravitreal biologic exists today
KSI-101 is a high-strength (100 mg/ml), locally administered, bispecific biologic designed to simultaneously target the two disease drivers of MESI – IL-6-mediated inflammation and VEGF-mediated vascular permeability
The unique promise of KSI-101 supported by emerging clinical data
Bispecific
KSI-101 targets both IL-6 and VEGF
Rapid and Powerful Drying Effect
In the Phase 1b APEX study in MESI, meaningful vision gains were rapidly achieved as early as Week 4. Over 90% of patients achieved retinal dryness by Week 8. Learn more
Favorable Early Clinical Safety Profile
In the Phase 1b APEX study in MESI, KSI-101 was well tolerated at all dose levels. The top two dose levels (5 mg and 10 mg) were selected to advance into the Phase 3 program. Learn more
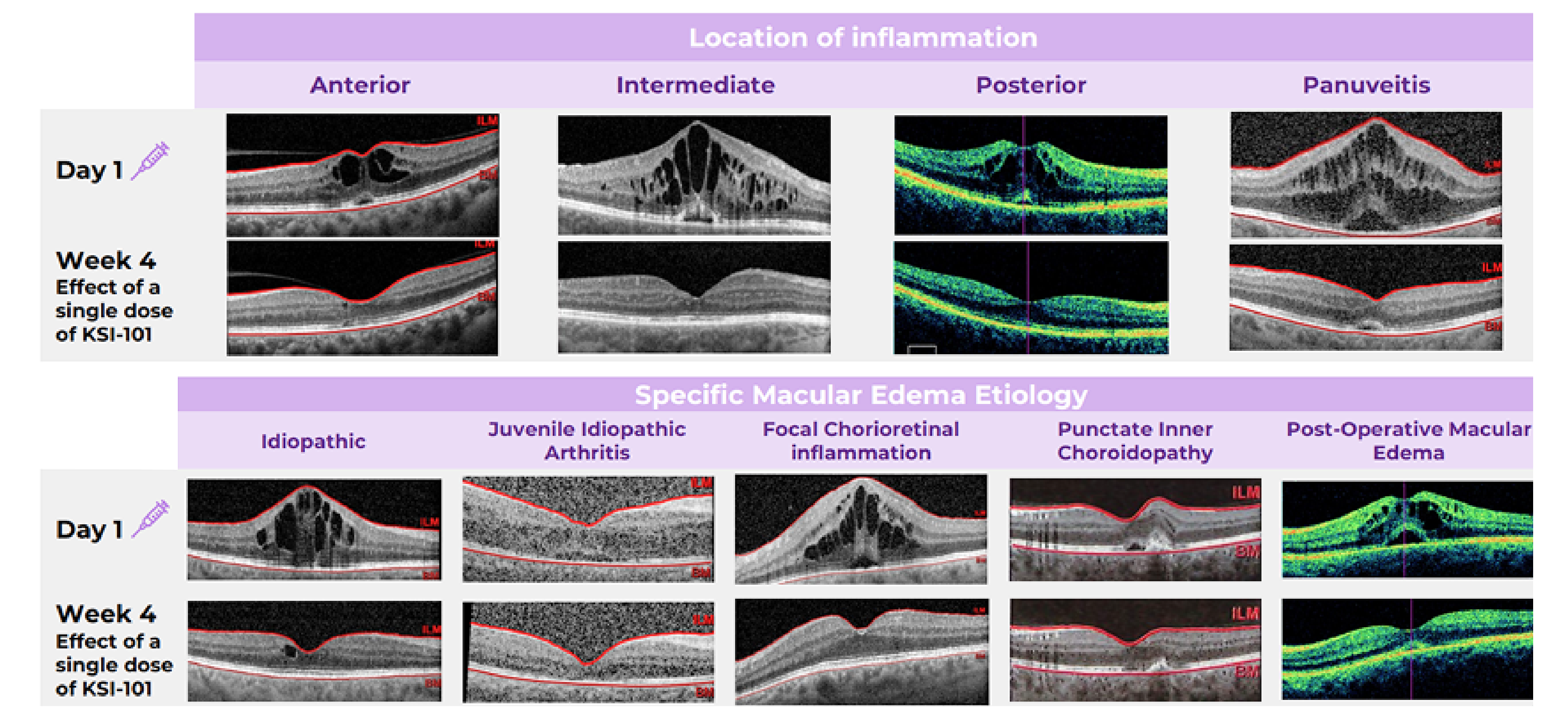
Broad Activity Across MESI Patients
KSI-101 demonstrated rapid and meaningful responses irrespective of the location of inflammation or the specific macular edema diagnosis. Learn more
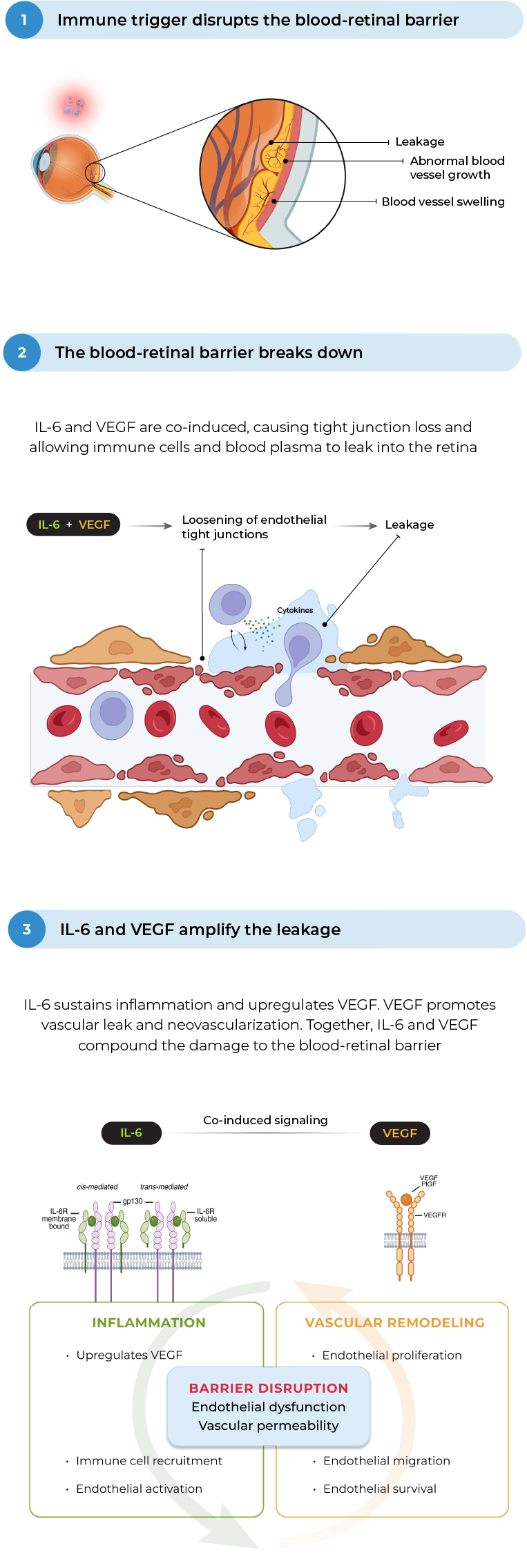
In MESI, IL-6 and VEGF act together to worsen damage to the blood-retinal barrier and increase vascular permeability
MESI occurs when an immune trigger (such as an autoimmune disease) disrupts the blood-retinal barrier, allowing immune cells and blood plasma into the retina, causing macular edema, visual impairment and worsening inflammation
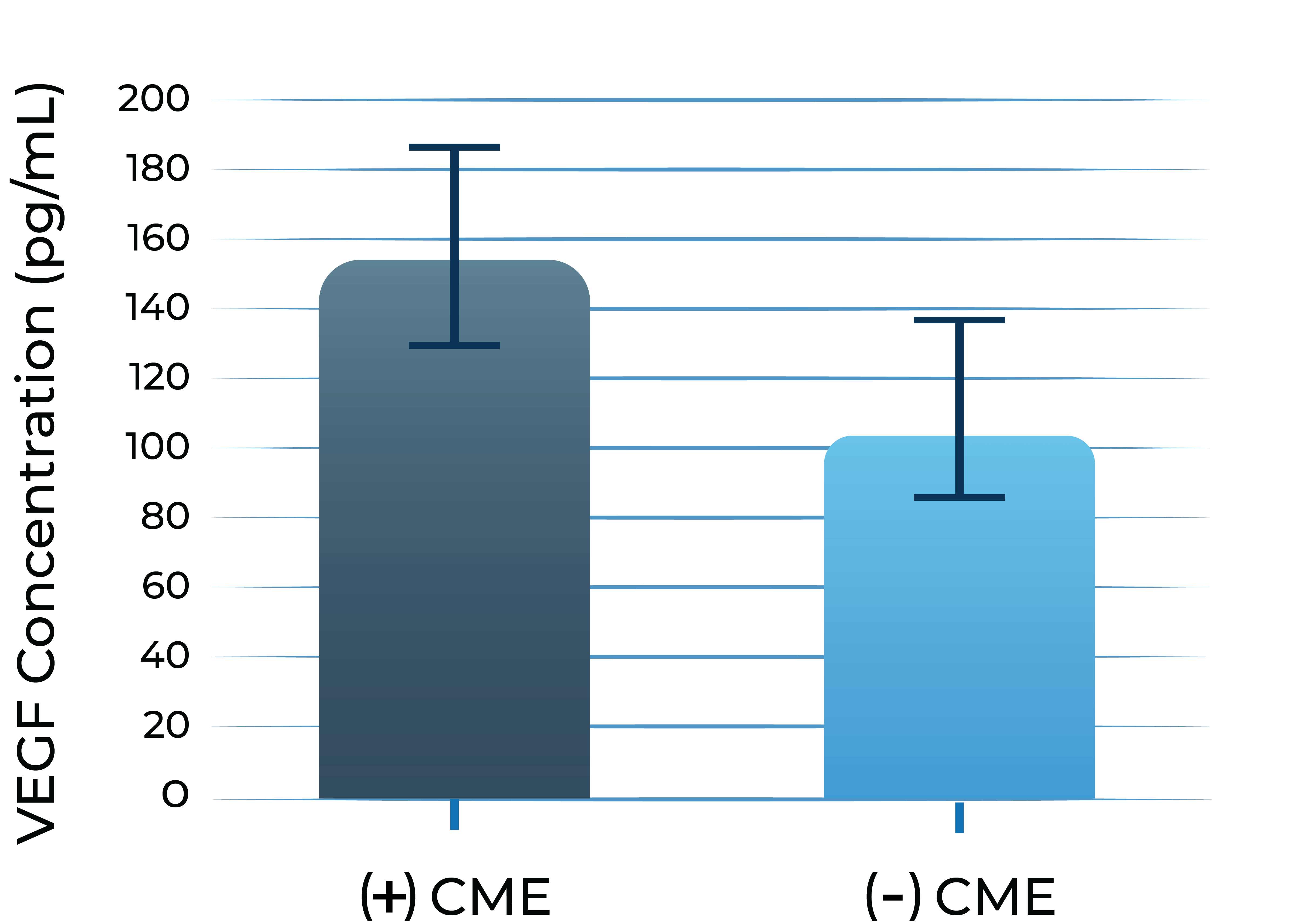
Both IL-6 and VEGF levels are elevated in inflammatory macular edema
Aqueous humor IL-6 levels were elevated in patients with intermediate uveitis
VEGF levels in aqueous humor of uveitis patients with macular edema vs without macular edema
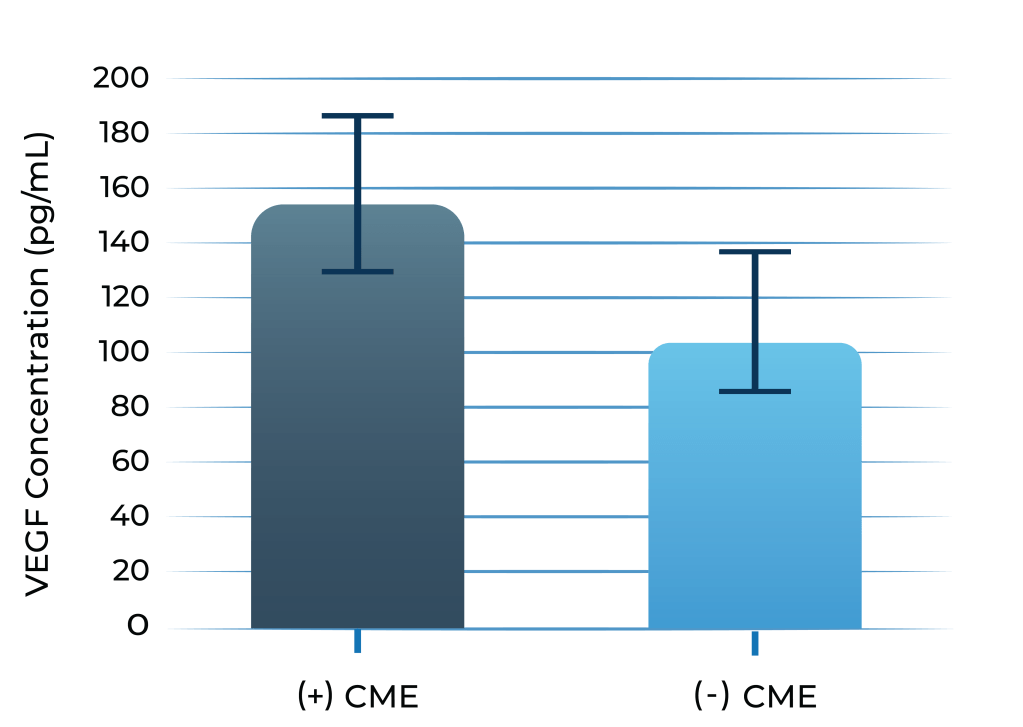
1. Valentincic et al. Molecular Vision 2011; 17: 2003-2010
2. de Boer et al. Curr Eye Res. 1992;11 Suppl:181-186
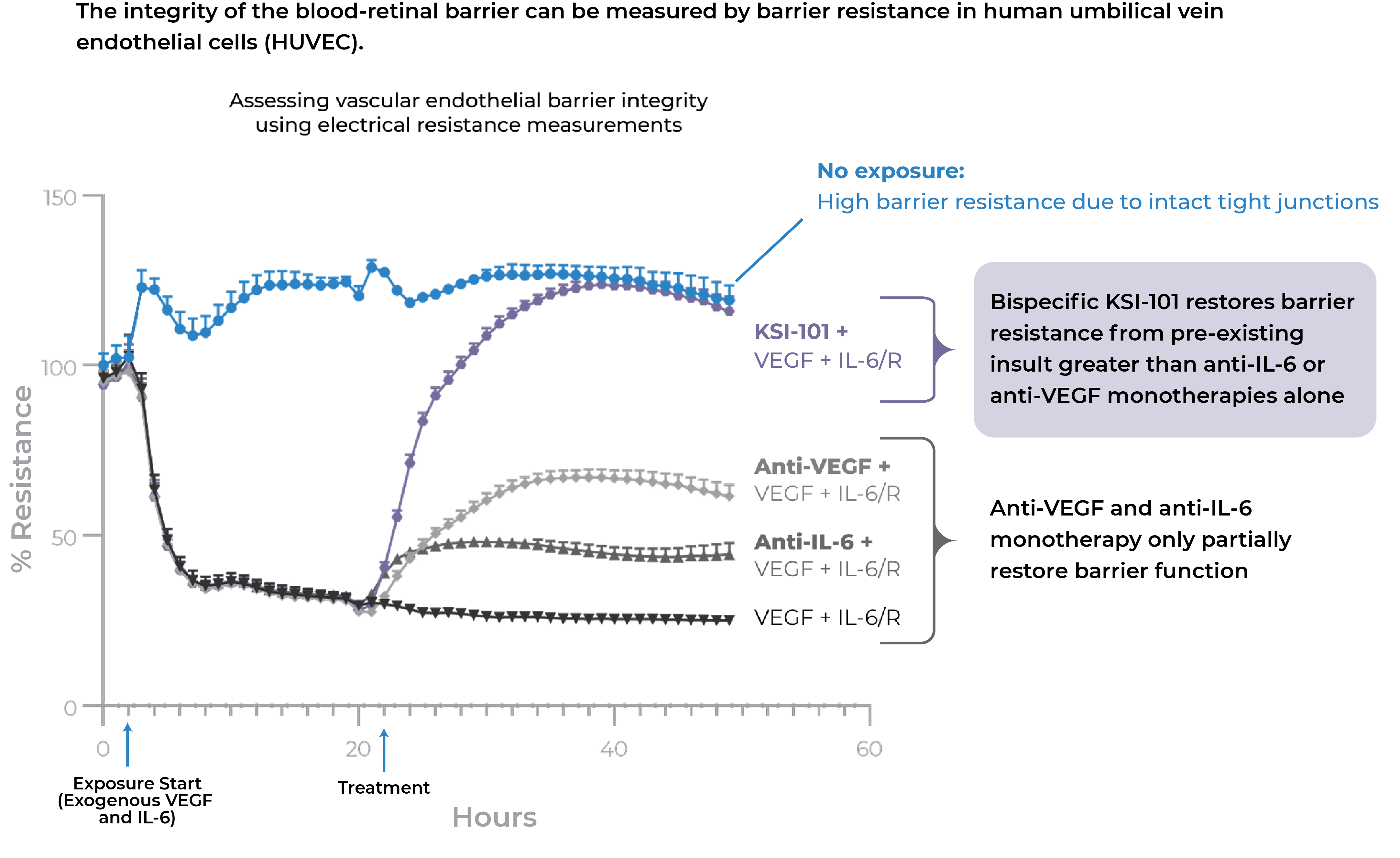
In a preclinical study, bispecific KSI-101 restores blood-retinal barrier integrity more effectively than IL-6 or VEGF inhibitors alone
In a preclinical model of endothelial cells simulating the blood-retinal barrier, bispecific KSI-101 restores blood-retinal barrier integrity following IL-6 and VEGF insult to a greater extent than anti-VEGF or anti-IL-6 monotherapy
Blood-retinal barrier exposed to VEGF and IL-6

KSI-101 is actively enrolling patients in the Phase 3 PEAK and PINNACLE studies
The APEX Phase 1b study demonstrates that KSI-101 provides meaningful visual and anatomical gains in both DME and MESI and that KSI-101 is well tolerated
See final APEX Phase 1b results
Meaningful treatment responses were seen in the MESI population, irrespective of the location of inflammation and specific MESI etiology, opening up the potential for KSI-101 to become a unifying treatment for this patient population
|
Phase 1b APEX Study Diabetic Macular Edema Macular Edema Secondary |
|
The KSI-101 program is accelerating based on the positive results of APEX. The Phase 3 PEAK and PINNACLE studies are actively enrolling. PEAK and PINNACLE are superiority studies designed to enroll complementary patient populations and to cover a wide spectrum of MESI patients
|
Phase 3 PEAK & PINNACLE Studies Macular Edema Secondary |
|
Multi-mechanism, multi-modality targeted biologic for complex retinal and systemic diseases
Triplet medicines combine the benefits of our Antibody Biopolymer Conjugate (ABC) Platform for long-interval dosing of biologics with a new feature that adds sustained release of 100s of small molecules to target three or more mechanisms of action, enabling treatment of complex, multifactorial diseases.
Designing a new generation of targeted therapy for high-prevalence multifactorial diseases

Our triplet ABC medicines aim to broaden what’s possible with antibody conjugate therapies
Antibody Drug Conjugate (ADC) therapies are revolutionizing the way cancer is treated today by delivering highly potent cancer-killing agents directly to cancer cells via a targeted antibody. With our ABC triplet medicines, we aim to build on this foundation in notable ways:
ADC
|
ABC Triplet Medicine
|
Our goal with our triplet medicines is to deliver greater therapeutic benefit for multifactorial diseases in the eye and systemically by modulating multiple distinct pathological processes in parallel



-04.png)